What is Pressure? What are the Pressure Units?
Pressure is commonly characterized as the force exerted on a specific region. It is a physical measure typically articulated using the equation p = F/A, where ‘F’ represents the force perpendicular to the surface, and ‘A’ denotes the surface area.
Units for Measuring Pressure:
Pressure receives measurement through a spectrum of units within different systems. Here, we explore frequently utilized units:
SI Unit (International System of Units):
Pascal ¶: The quintessential SI unit for pressure. Its subdivisions encompass mPa (millipascal) and kPa (kilopascal), catering to meticulous scientific and engineering contexts.
1 MPa = 1,000 kPa = 1,000,000 Pa
CGS Unit (Centimeter-Gram-Second System):
Barye (Ba): Less pervasive, this unit equals 1 dyne per square centimeter (dyn/cm²) within the CGS system.
Imperial Units:
Pounds per Square Inch (psi): Prominently embraced in the United States, signifying the pressure arising from a one-pound force applied over a one-square-inch surface.
Alternative Units:
Atmosphere (atm): In widespread use, particularly at sea level, where the established atmospheric pressure approximates 101,325 Pa or 1 atm.
Torr: Tied to 1 mmHg (millimeter of mercury), named in homage to Evangelista Torricelli.
Bar: Equating to 100,000 Pa.
Technical Atmosphere (at): Find utility in specific domains, defined as 1 kgf/cm².
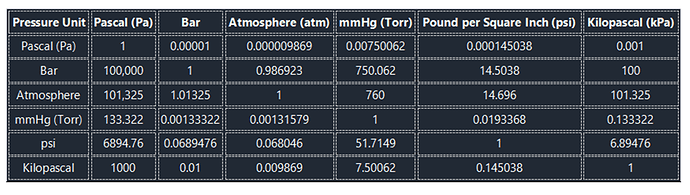
The following is a table for easy and direct conversion between different pressure units.